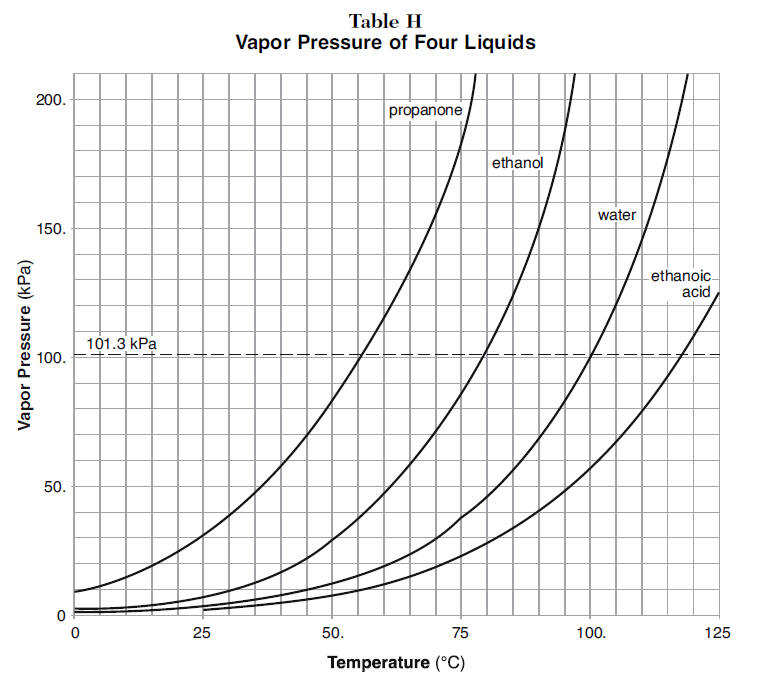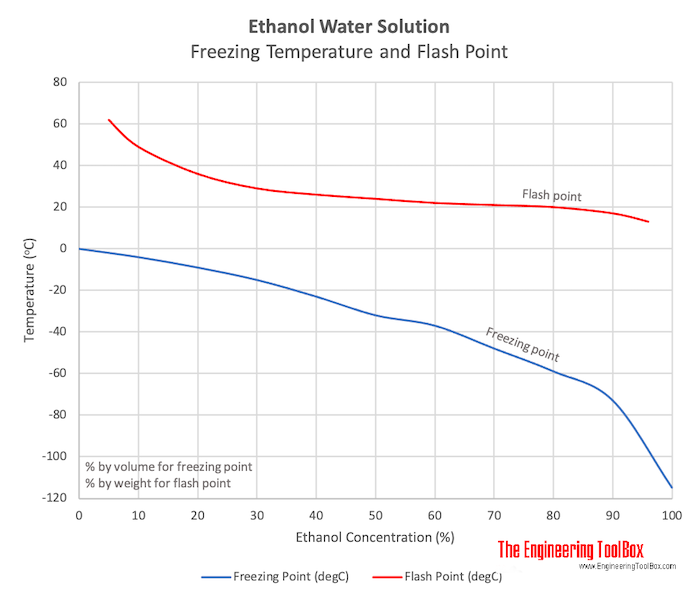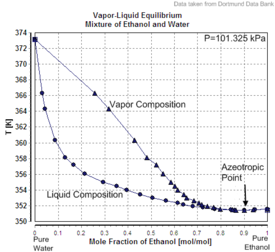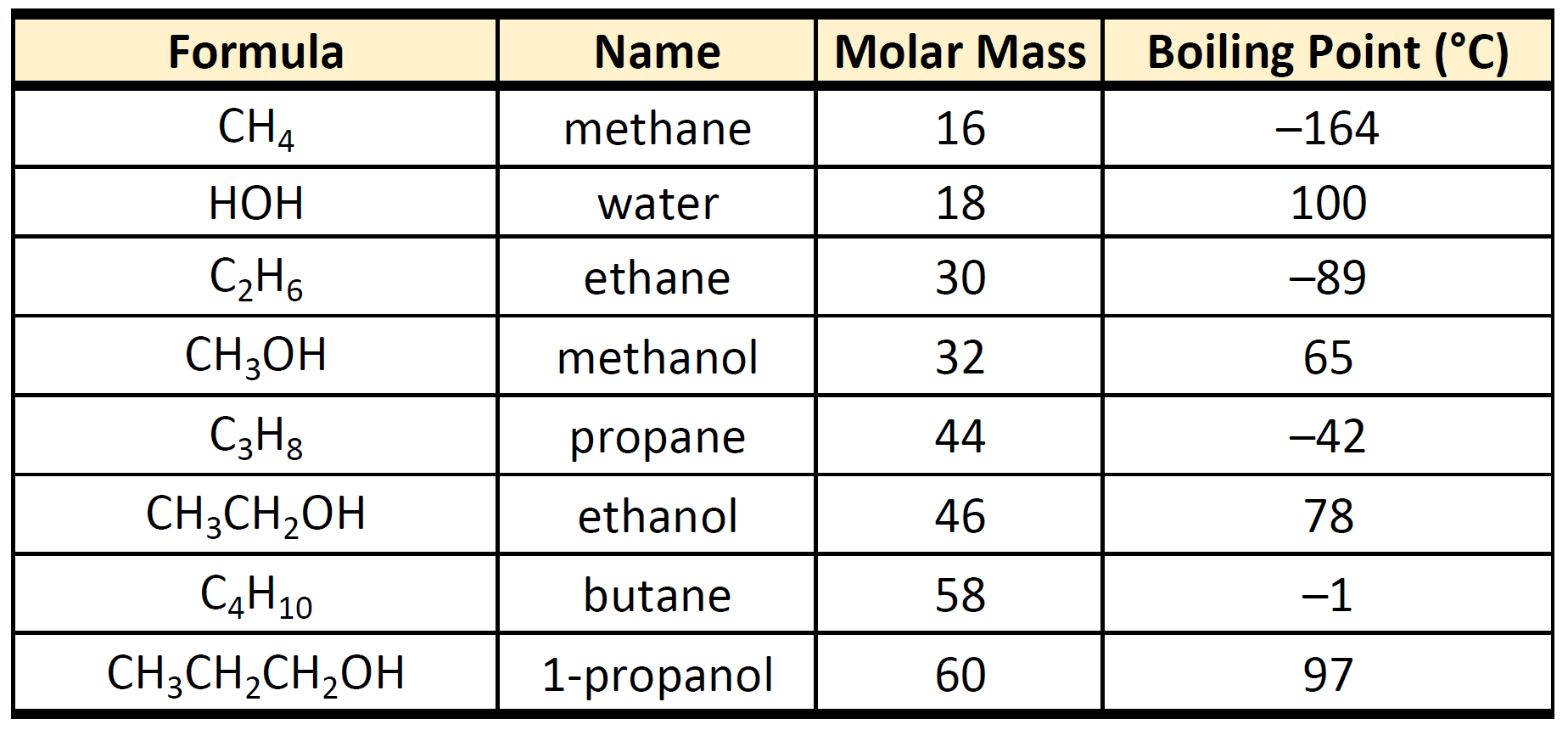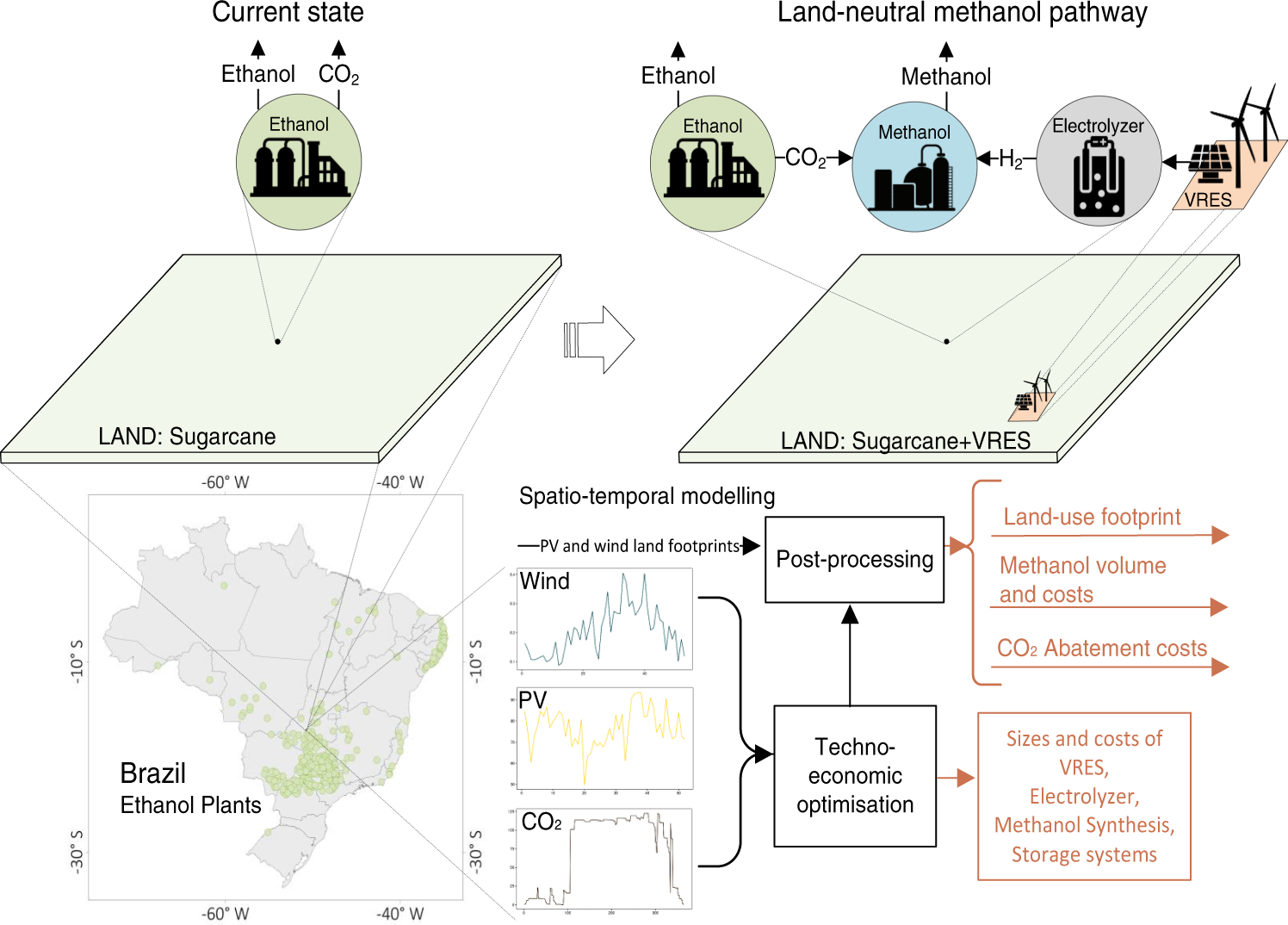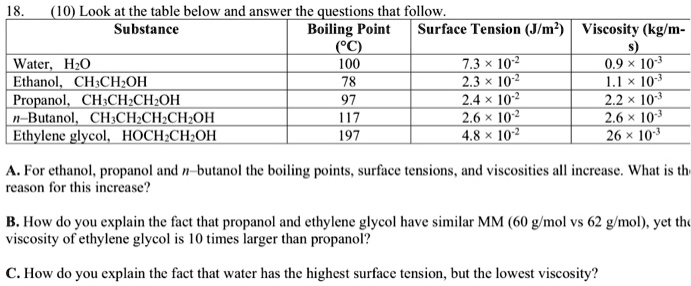
SOLVED: (1Q) Look at the table below and answer the questions that follow Substance Boiling Point Surface Tension (J/m ) Viscosity (kglm Water. HzO Ethanol;. CHCHOH Propanol. CHCHCHOH Butanol. CHCHCHCHOH Ethylene glycol,
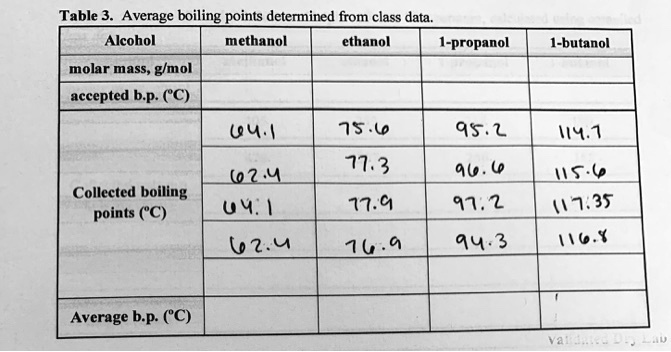
SOLVED: Table 3. Average boiling points determined from class data Alcohol methanol ethanol 1-propanol molar mass, glmol accepted bp CC) F-butanol 64; 75 95.2 1/4.1 (0.24 04; | 77.3 17.4 9b. 6
a) Equilibrium phase diagrams of water, cyclohexane, and ethanol and b)... | Download Scientific Diagram

Question Video: Separating and Collecting Four Liquids That Are Mixed Together Depending on Their Boiling Point Using the Fractional Distillation Technique | Nagwa
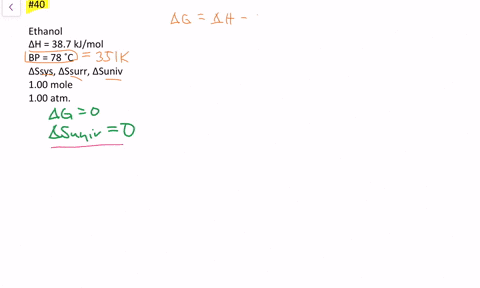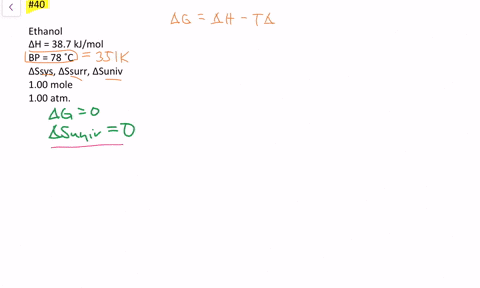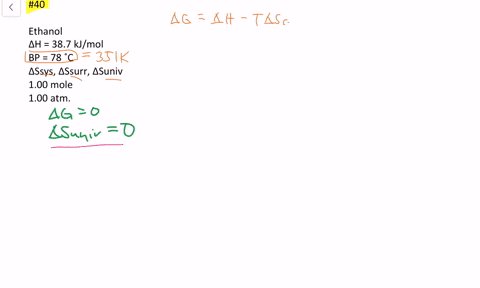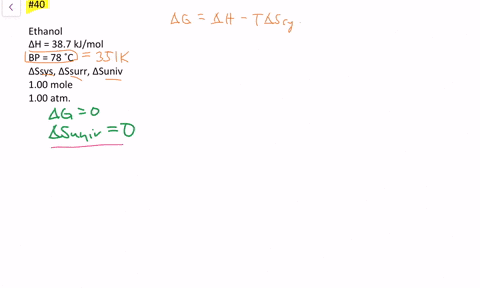
SOLVED:The enthalpy of vaporization of ethanol is 38.7 kJ / mol at its boiling point (78^∘ C) . Determine ΔSsys, ΔSsurr and ΔSuniv when 1.00 mole of ethanol is vaporized at 78^∘ C and 1.00 atm.

Observed (data points, with ethanol and oleate microcosm test data from... | Download Scientific Diagram